⚗️
Co-Formulated Peptide Stacks for Sale
Pre-formulated peptide co-formulations — BPC-157/TB-500, Cagri-Sema, Ipa/CJC, and metabolic multi-receptor stacks.
In this category · 8 peptides
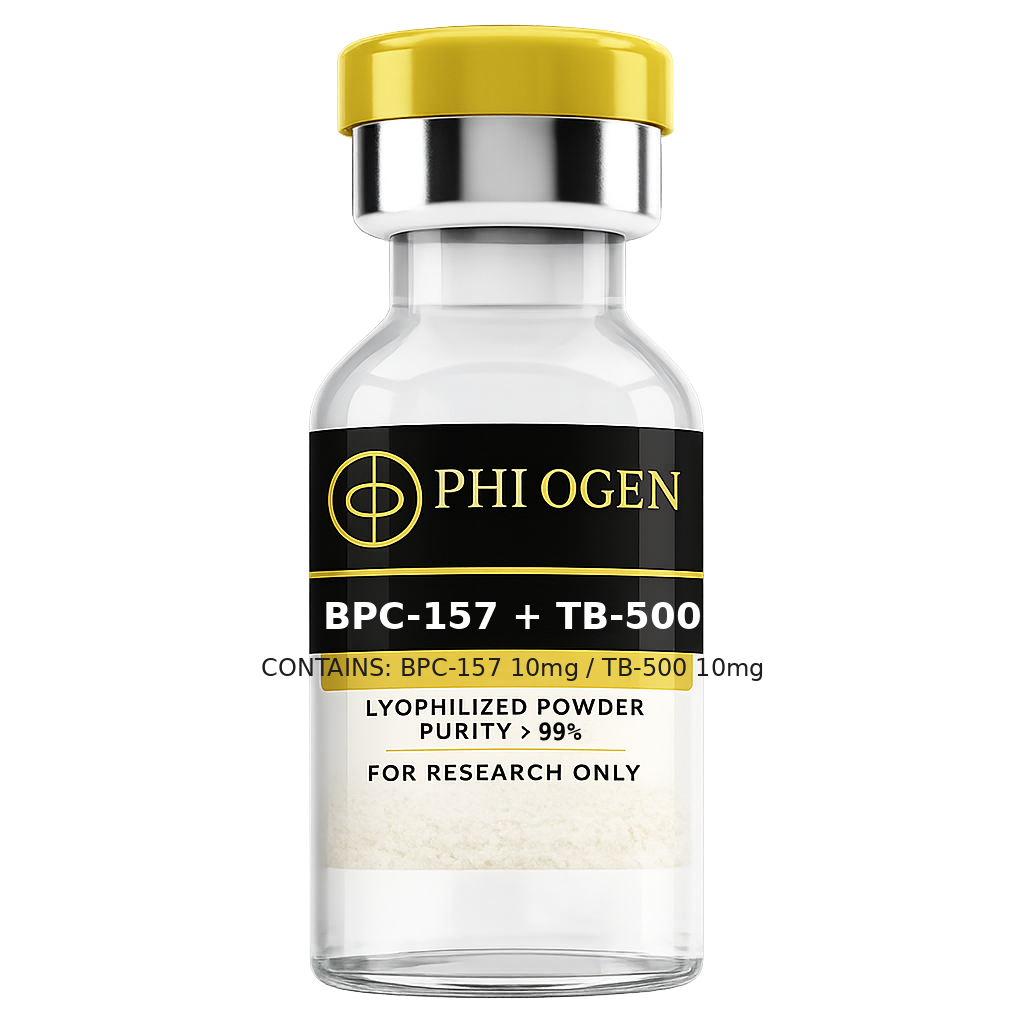
Blend
2 sizes
BPC-157 + TB-500 Blend
From
$53.99

Blend
2 sizes
Cagri-Reta Blend
From
$116.99

Blend
Cagri-Sema Blend
From
$161.99

Blend
2 sizes
GLOW Blend
From
$80.99
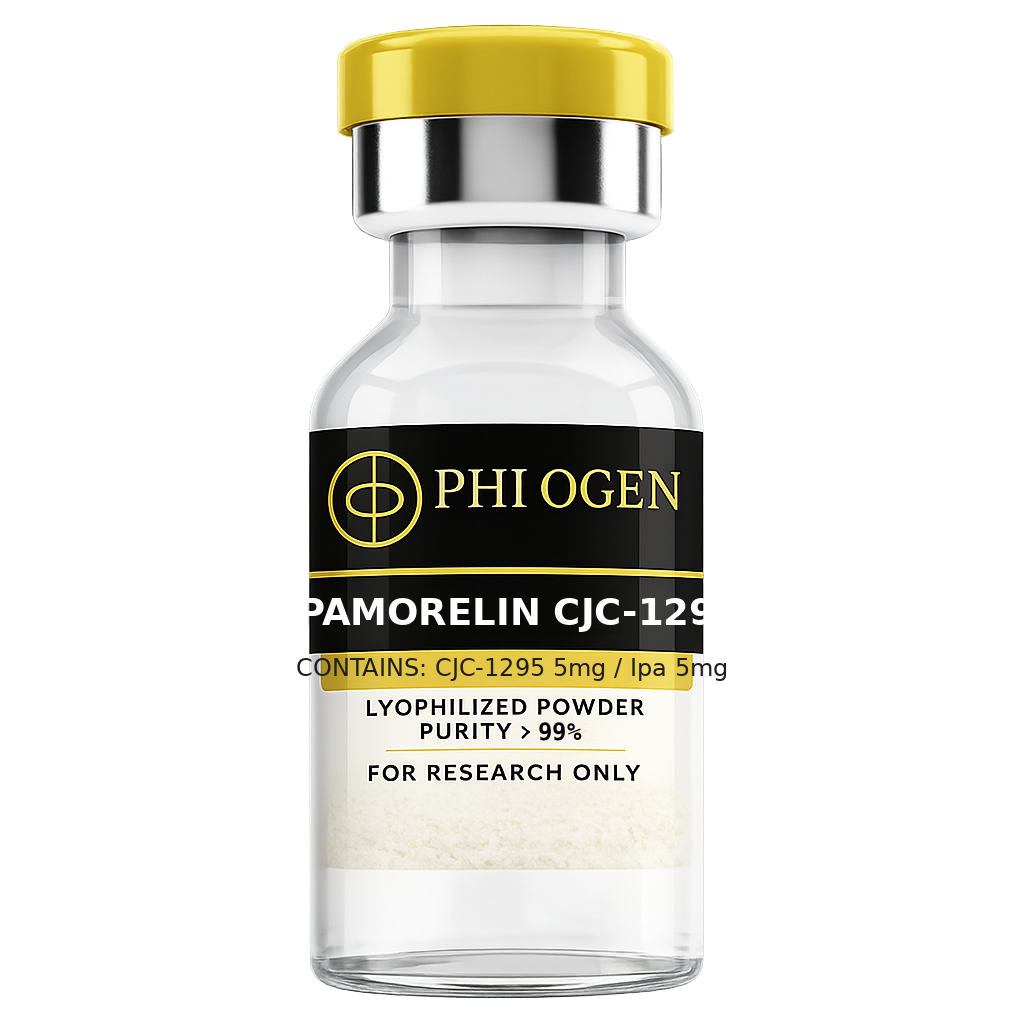
Blend
2 sizes
Ipamorelin/CJC-1295 Blend
From
$80.99

Blend
3 sizes
Ipamorelin/Tesamorelin Blend
From
$116.99

Blend
KLOW Blend
From
$107.99

Blend
Reta-Cagri Blend
From
$134.99
Need a different size or blend? Open any product for pricing, purity, shipping windows, and storage notes. Everything on the shelf is sold for laboratory use only — not for human consumption.
Featured in this category
Compliance
Laboratory use only. Not for human consumption, injection, or resale as a finished drug. Check local rules before ordering.